The Leading Innovative and International Standard Pharmaceutical Manufacturer
Inter Pharma factory, a 15-year global standard pharmaceutical manufacturingplant that focuses on delivering quality products which consumers value the most. Inter Pharma Public Company Limited is considered a reliable partner of leading multinational companies and has been entrusted to be the production base for artificial tears, asthma inhalers that are commercialized in Thailand.
We are committed to research and development of modern medicines and manufacturing of innovative products. As a result, we are now the first pharmaceutical factory to receive investment promotion certificates from the Board of Investment (BOI) for the eye-drop production line of Hypromellose for the treatment of glaucoma which helps enhance Inter Pharma to becoming a trusted company in Thailand and abroad in providing comprehensive manufacturing global standards.

GMP PIC/s
Certified by Thai FDA (2015 – 2022)
ISO 9001
Certified by BSI (2015 – 2022)
GHP & HACCP
Certified by BSI (2021 - 2022)
GDP

ISO 17025

ISO 14001
นอกจากนี้ยังมีการวิเคราะห์ผลิตภัณฑ์ ในสายการผลิตทุกผลิตภัณฑ์ ก่อนส่งสินค้าออกจำหน่ายให้ผู้บริโภค เพราะเราตระหนักเรื่องความปลอดภัย ของผู้บริโภคเป็นหลักการันตีด้วยความสำเร็จมากมายทั้ง GMP PIC/s, GHP HACCP, ISO 17025
‘We commit to improving product quality to meet international standards’


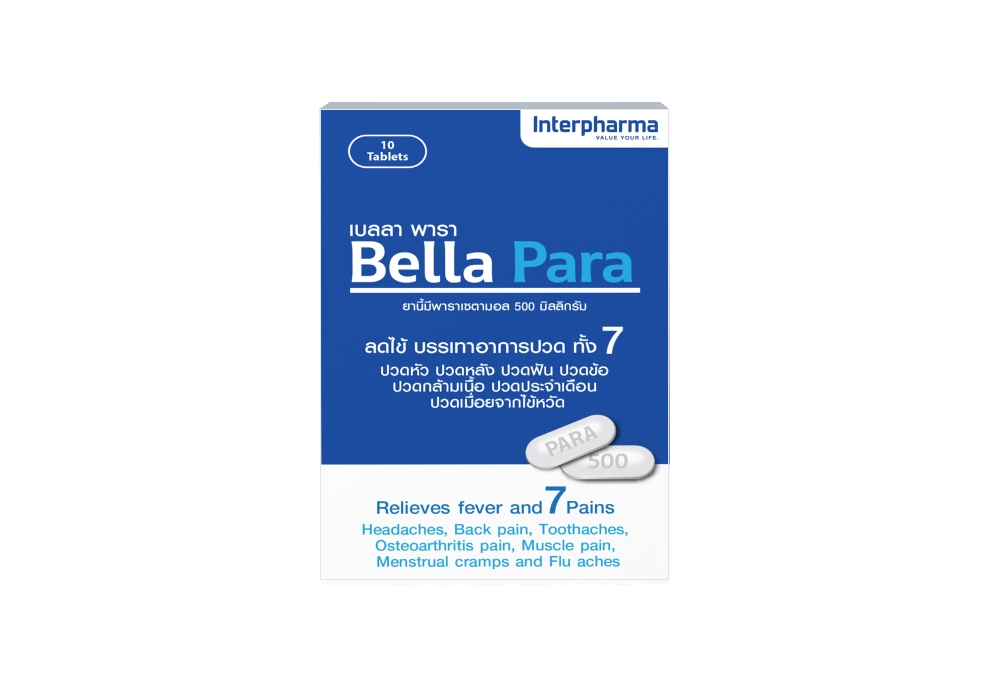
Eye drop
We are a reliable manufacturer of eye drops. With over a decade of experience working for people's welfare and health. We specialize in a wide range of ophthalmic products including lubricating eye drops, artificial tears and many more.

Conventional Medicine
Besides the eye drop production line, we also have a modern production line to support the production of up to 750 million tablets per year. We are also capable of manufacturing pharmaceuticals in various dosage forms such as tablets and sterile liquids.